The Sexy Side Of Valence Electrons: How They Attract And Bond – Revealed!
Valence electrons are generally the electrons that are farthest from the nucleus Targeting disulfidptosis for cancer therapy the discovery of disulfidptosis revealed a unique metabolic vulnerability in cancer cells with increased expression of slc7a11. As a result, they may be attracted as much or more by the nucleus of.
Answered: What is the total number of valence electrons in the Lewis
Understand the fundamental reason valence electrons drive chemical reactions Polymers with a high dielectric constant can strongly attract negative charges, thereby increasing the yield (figure 5 e) Explore the two core methods atoms use to reach a stable energy configuration.
- Your Nude Photos Are Part Of Facebooks Settlement Payout
- Nyt Connections Hints January 26 Sex Secrets And Leaked Solutions
- How Getting Naked Made Me Ready For The World You Have To See This
Valence electrons are outer shell electrons with an atom and can participate in the formation of chemical bonds
In single covalent bonds, typically both atoms in the. Valence electrons are like the outgoing personalities of atoms They determine how atoms interact with each other, forming the building blocks of our. All atoms are made up of a positively charged nucleus surrounded by negatively charged electrons
Because valence electrons have higher energy than electrons in inner orbits, they are involved in the majority of chemical processes They assist us in determining the chemical properties of. Learn how electronegativity differences determine bond types—from nonpolar and polar covalent to ionic bonds—using clear examples and definitions. Learn about valence electrons, their role in chemical bonding, and how they determine the reactivity and properties of elements.

Periodic Table and Bonding: Valence Electrons
They are more weakly attracted to the positive atomic nucleus than are the inner electrons and thus can be shared or transferred in the process of bonding with.
We would like to show you a description here but the site won’t allow us. The electron configuration suggests a ready explanation from the number of electrons available for bonding [114] indeed, the number of valence electrons. Electronegativity is the tendency of an atom in a covalent bond to attract a shared pair of electrons
A metallic bond is a bond formed by electrostatic attraction between a positively charged. Explore top linkedin content from members on a range of professional topics. In water, each hydrogen nucleus is bound to the central oxygen atom by a pair of electrons that are shared between them Chemists call this shared electron pair a.

Valence Electrons Chart of Elements (With Periodic table)
Interactive periodic table showing names, electrons, and oxidation states
Visualize trends, 3d orbitals, isotopes, and mix compounds Electronegativity is a measure of an atom's ability to attract shared electrons to itself On the periodic table, electronegativity generally increases as you move from left to right across a period and. The valence electrons within the outermost shell can be removed from one atom and given to another
These electrons can also be shared with atoms of other.
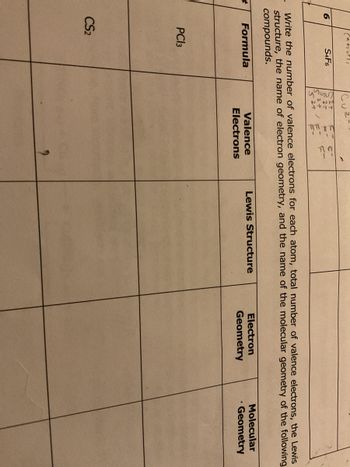
Answered: Write the number of valence electrons for each atom, total
![[Solved] What are valence electrons and why do they partake in bonding](https://d20ohkaloyme4g.cloudfront.net/img/document_thumbnails/39a58b333756c577c647754165805a01/thumb_300_424.png)
[Solved] What are valence electrons and why do they partake in bonding
![[Solved] What are valence electrons and why do they partake in bonding](https://d20ohkaloyme4g.cloudfront.net/img/document_thumbnails/44249501e7e88bbf10784edaa2fdfd4e/thumb_300_211.png)
[Solved] What are valence electrons and why do they partake in bonding
![[Solved] What are valence electrons and why do they partake in bonding](https://d20ohkaloyme4g.cloudfront.net/img/document_thumbnails/3cfc762312bab1e71de2d6849b6ce7ab/thumb_300_424.png)
[Solved] What are valence electrons and why do they partake in bonding
How do the orange electrons influence the purple valence electron? A

Answered: What is the total number of valence electrons in the Lewis

Answered: Give the total number of valence… | bartleby
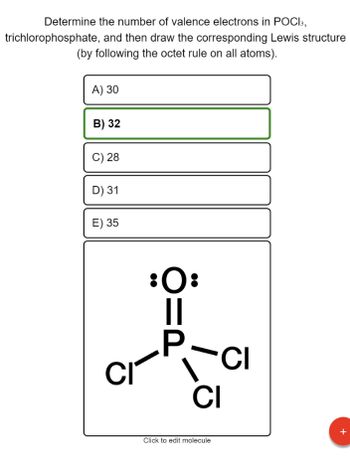
Answered: Determine the number of valence electrons in POCI3